In the history of medical science, few intellectual revolutions have transformed our understanding of disease and healing as profoundly as the emergence of regenerative medicine and regenerative pharmacology. Traditional medicine has long focused on controlling symptoms, suppressing disease progression, or replacing damaged organs through surgery or transplantation. However, a new paradigm is rapidly emerging—one that seeks not merely to treat disease but to restore, repair, and regenerate damaged tissues and organs at their biological roots. Regenerative pharmacology and regenerative medicine represent a powerful convergence of molecular biology, pharmacology, biotechnology, stem cell science, and tissue engineering, offering humanity an unprecedented opportunity to rethink the nature of healing itself.
The promise of regeneration is deeply embedded in nature. Certain organisms, such as salamanders and starfish, possess remarkable regenerative capacities, allowing them to regrow entire limbs or organs after injury. For centuries, scientists have been fascinated by this phenomenon, attempting to understand whether similar regenerative capabilities could be harnessed within the human body. The emergence of regenerative medicine marks the scientific translation of this ancient curiosity into practical therapeutic strategies capable of transforming modern healthcare systems.
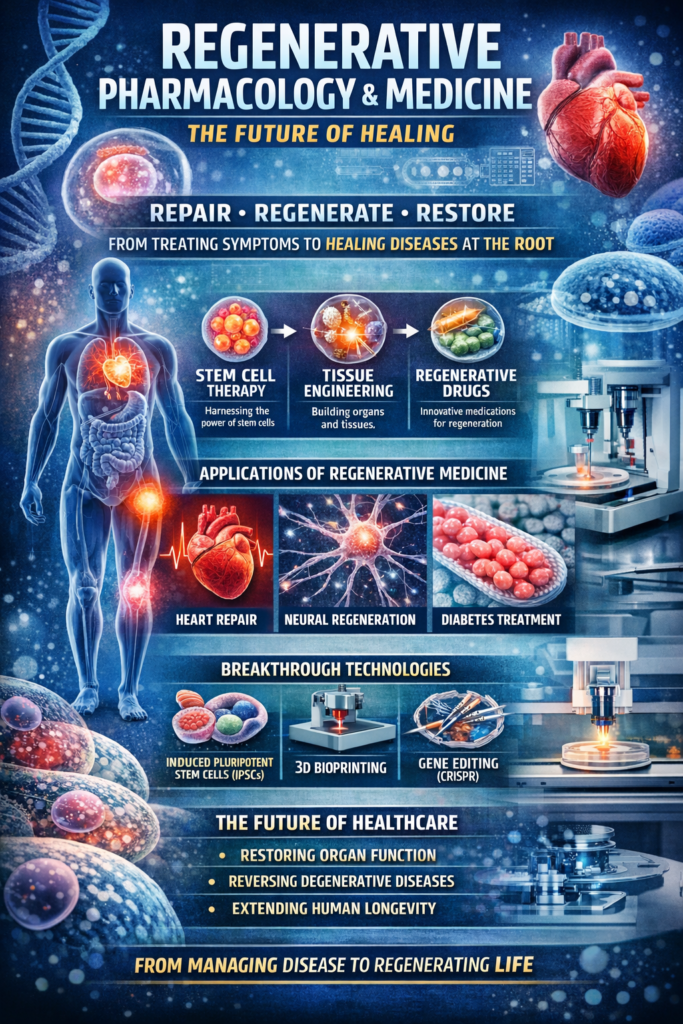
Regenerative medicine is fundamentally defined as a branch of biomedical science focused on repairing, replacing, or regenerating human cells, tissues, or organs to restore normal biological function. It represents a transition from reactive medicine toward restorative medicine. Instead of managing chronic diseases indefinitely, regenerative medicine aims to eliminate their biological origins by rebuilding damaged tissues. Regenerative pharmacology, a closely related discipline, explores pharmacological agents that stimulate, modulate, or enhance regenerative biological processes within the body.
The scientific foundations of regenerative medicine were laid during the late twentieth century with the discovery of stem cells. Stem cells are unique biological entities capable of self-renewal and differentiation into specialized cell types. These cells possess the ability to generate new tissues, making them central to regenerative therapies. Stem cell research has revealed that under appropriate conditions, these cells can be directed to develop into cardiac cells, neurons, skin cells, or even pancreatic beta cells. Such discoveries have opened new therapeutic pathways for diseases previously considered irreversible.
Regenerative pharmacology expands this concept by developing pharmacological molecules that can activate or regulate these regenerative processes. Unlike traditional drugs that simply block biochemical pathways, regenerative pharmacological agents attempt to stimulate cellular renewal, promote tissue repair, and restore physiological balance. This emerging discipline integrates classical pharmacodynamics with advanced cellular biology to create drugs that influence stem cell activity, growth factor signaling, extracellular matrix remodeling, and tissue regeneration.
One of the central pillars of regenerative medicine is stem cell therapy. Stem cells can be broadly categorized into embryonic stem cells, adult stem cells, and induced pluripotent stem cells. Embryonic stem cells possess pluripotent capabilities, meaning they can differentiate into nearly any cell type in the body. However, ethical controversies surrounding their use have limited their widespread clinical application. Adult stem cells, such as hematopoietic stem cells found in bone marrow, have been successfully used for decades in the treatment of blood disorders through bone marrow transplantation.
The discovery of induced pluripotent stem cells revolutionized the field of regenerative medicine. These cells are created by reprogramming adult somatic cells into pluripotent states, effectively converting ordinary cells into stem-like cells capable of generating diverse tissues. This breakthrough eliminated many ethical concerns associated with embryonic stem cells and opened new possibilities for personalized regenerative therapies.
Another important component of regenerative medicine is tissue engineering. Tissue engineering combines biomaterials, cells, and bioactive molecules to create functional tissues that can replace damaged biological structures. Researchers have successfully engineered skin grafts, cartilage tissues, and even experimental heart valves using sophisticated biomaterial scaffolds. These scaffolds serve as structural frameworks that support cellular growth and guide tissue formation.
Biomaterials used in tissue engineering are carefully designed to mimic the natural extracellular matrix, the complex environment surrounding cells within living tissues. Advanced biomaterials can release growth factors, provide mechanical support, and promote cell adhesion, proliferation, and differentiation. The integration of biomaterials with stem cell biology has enabled scientists to design tissue constructs capable of repairing complex tissue injuries.
Regenerative pharmacology plays a critical role in supporting these tissue engineering approaches. Pharmacological agents such as growth factors, cytokines, and signaling modulators are used to regulate cellular behavior within engineered tissues. For example, vascular endothelial growth factor (VEGF) stimulates the formation of new blood vessels, a process known as angiogenesis. This is essential for ensuring that engineered tissues receive adequate oxygen and nutrients after transplantation.
One of the most exciting technological advancements in regenerative medicine is three-dimensional bioprinting. Bioprinting involves the use of specialized printers that deposit living cells and biomaterials layer by layer to create complex tissue structures. Scientists have successfully bioprinted skin tissues, cartilage, and experimental organ prototypes using this technology. Although fully functional organ printing remains an ambitious goal, rapid progress in biomaterials science and cellular engineering suggests that it may become a clinical reality within the coming decades.
Regenerative pharmacology is also deeply connected to the concept of endogenous regeneration. Rather than introducing external cells or tissues, endogenous regeneration focuses on stimulating the body’s own repair mechanisms. Certain tissues, such as the liver and skin, already possess strong regenerative capabilities. Pharmacological agents can potentially amplify these natural processes, enabling damaged organs to repair themselves more efficiently.
For example, regenerative pharmacological strategies are being explored to stimulate heart tissue regeneration after myocardial infarction. Traditionally, heart muscle cells lost during a heart attack are permanently replaced by scar tissue, leading to reduced cardiac function. However, researchers are developing molecules that activate dormant cardiac progenitor cells or promote cardiomyocyte proliferation, potentially enabling the heart to regenerate damaged tissue.
Neuroregeneration represents another promising frontier in regenerative medicine. The central nervous system has historically been considered incapable of significant regeneration. However, advances in stem cell biology and neuropharmacology suggest that certain neural stem cells may be capable of generating new neurons under specific conditions. Regenerative pharmacology is exploring molecules that stimulate neural growth, repair synaptic connections, and promote recovery after spinal cord injury or neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease.
Regenerative medicine also holds transformative potential for the treatment of diabetes. Type 1 diabetes is characterized by the destruction of insulin-producing beta cells in the pancreas. Regenerative therapies aim to restore insulin production by generating new beta cells from stem cells or by stimulating pancreatic regeneration. Clinical trials involving stem cell-derived pancreatic cells are currently underway in several countries.
Another critical area of regenerative medicine is orthopedic regeneration. Conditions such as osteoarthritis, cartilage injuries, and bone fractures often require surgical interventions or prosthetic implants. Regenerative therapies using stem cells, growth factors, and biomaterial scaffolds can potentially regenerate cartilage and bone tissues, reducing the need for joint replacement surgeries.
Regenerative pharmacology also intersects with gene therapy and molecular medicine. Gene editing technologies such as CRISPR-Cas9 allow scientists to modify genetic material within cells, correcting mutations responsible for inherited diseases. When combined with regenerative approaches, gene editing can enable the creation of healthy cells capable of replacing defective tissues.
The pharmaceutical industry is increasingly recognizing the transformative potential of regenerative pharmacology. Traditional drug development has often focused on small-molecule drugs that target specific biochemical pathways. However, regenerative pharmacology requires a broader therapeutic framework that includes biologics, gene therapies, cell-based treatments, and advanced biomaterials.
This shift represents a profound transformation in pharmaceutical innovation. The regulatory landscape is also evolving to accommodate these complex therapies. Regulatory agencies such as the U.S. Food and Drug Administration and the European Medicines Agency have established specialized pathways for regenerative medicine products, recognizing their unique scientific and manufacturing challenges.
Despite its enormous promise, regenerative medicine faces significant scientific, ethical, and economic challenges. One major concern is the risk of uncontrolled cell growth, which could potentially lead to tumor formation. Ensuring the safety and stability of stem cell-derived therapies remains a critical area of research.
Ethical considerations also play an important role in the development of regenerative technologies. The use of embryonic stem cells has generated intense ethical debates in many countries. While induced pluripotent stem cells have alleviated some of these concerns, questions regarding genetic manipulation and long-term safety remain subjects of ongoing discussion.
Economic challenges are equally significant. Regenerative therapies are often highly complex and expensive to develop and manufacture. The cost of advanced cell therapies can reach hundreds of thousands of dollars per treatment, raising concerns about healthcare accessibility and affordability.
Nevertheless, the global regenerative medicine market is expanding rapidly. Governments, biotechnology companies, and academic institutions are investing heavily in research and development. International collaborations are accelerating scientific discoveries, bringing regenerative therapies closer to clinical reality.
For countries like India, regenerative pharmacology represents both an opportunity and a strategic necessity. India possesses a strong pharmaceutical manufacturing base, a rapidly growing biotechnology sector, and a large pool of skilled scientific professionals. By investing in regenerative medicine research, India can position itself as a global leader in next-generation healthcare innovation.
Indian institutions such as the Indian Council of Medical Research and the Department of Biotechnology have already initiated programs supporting stem cell research and regenerative medicine. However, greater integration between academia, industry, and government policy will be essential to fully realize the potential of regenerative technologies in the Indian healthcare ecosystem.
Regenerative pharmacology also has profound implications for the future of human longevity. As regenerative therapies mature, it may become possible to repair age-related tissue damage, delay degenerative diseases, and extend healthy human lifespan. While such possibilities raise philosophical and societal questions, they also represent one of the most exciting scientific frontiers of the twenty-first century.
The ultimate vision of regenerative medicine is the creation of a healthcare system capable of restoring biological integrity rather than merely managing disease. In such a future, organ failure may be treated by regenerating new tissues, neurodegenerative disorders may be reversed through neural regeneration, and chronic diseases may be eliminated through cellular repair.
In conclusion, regenerative pharmacology and regenerative medicine represent a revolutionary transformation in biomedical science. By integrating stem cell biology, pharmacology, biomaterials engineering, and molecular medicine, these disciplines are redefining the boundaries of therapeutic possibility. While significant scientific and ethical challenges remain, the progress achieved over the past two decades demonstrates that regenerative medicine is no longer a distant dream but an emerging reality.
The journey toward regenerative healing reflects humanity’s enduring aspiration to overcome disease and restore health. As research continues to advance, regenerative pharmacology may ultimately reshape modern medicine, ushering in an era where healing is not limited to symptom management but extends to the regeneration of life itself.
