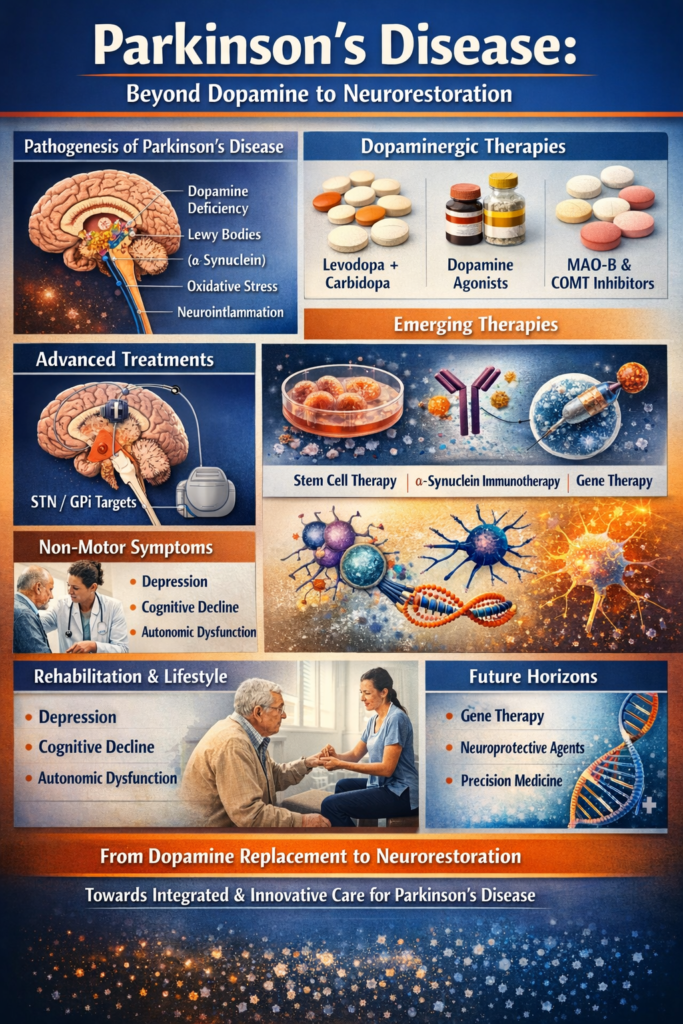
A Disorder of Movement, A Crisis of Identity
Parkinson’s disease (PD) is often reduced to tremor, rigidity, and slowness. Yet, this reductionist framing obscures its deeper reality. Parkinson’s disease is not merely a motor disorder; it is a progressive neurodegenerative syndrome that challenges the integrity of human autonomy, cognition, mood, and identity.
In a nation like India, where demographic transition is accelerating and life expectancy is steadily increasing, the burden of neurodegenerative disease is poised to expand exponentially. Parkinson’s disease thus becomes not only a neurological challenge but a public health imperative.
From a pharmacological standpoint, PD represents one of the most instructive examples of translational neuroscience: the journey from pathophysiological insight to therapeutic intervention has been remarkable—yet incomplete.
This article systematically explores treatment strategies for Parkinson’s disease in a structured, analytical–narrative framework: from classical dopaminergic replacement to advanced neuromodulation and emerging disease-modifying paradigms.
Background: Pathobiology and Therapeutic Logic
At its core, Parkinson’s disease is characterized by progressive degeneration of dopaminergic neurons in the substantia nigra pars compacta. The consequent dopamine deficiency in the striatum disrupts basal ganglia circuitry, producing the classical motor features:
- Resting tremor
- Bradykinesia
- Rigidity
- Postural instability
However, pathology extends far beyond dopamine. α-synuclein aggregation (Lewy bodies), mitochondrial dysfunction, oxidative stress, neuroinflammation, lysosomal impairment, and genetic susceptibility all contribute to disease progression.
This multifactorial pathology explains a central therapeutic dilemma:
Dopamine replacement corrects symptoms, but it does not arrest neurodegeneration.
Thus, treatment strategies must be viewed across three tiers:
- Symptomatic dopaminergic restoration
- Circuit modulation and motor fluctuation management
- Disease-modifying and neurorestorative approaches
I. Dopaminergic Replacement Therapy: The Foundational Pillar
Levodopa: The Gold Standard
No discussion of Parkinson’s treatment can begin without Levodopa.
Levodopa, a metabolic precursor of dopamine, crosses the blood–brain barrier via large neutral amino acid transporters and is decarboxylated to dopamine in surviving nigrostriatal neurons.
To prevent peripheral metabolism and reduce side effects, it is co-administered with Carbidopa or Benserazide.
Clinical Strengths
Most effective agent for bradykinesia and rigidity
Rapid onset of motor improvement
Improves quality of life dramatically
Limitations
Motor fluctuations (“wearing-off” phenomenon)
Dyskinesias with chronic exposure
Short plasma half-life
Does not modify disease progression
The paradox of levodopa therapy lies in its brilliance and its limitation: it restores movement but cannot restore neurons.
Dopamine Agonists: Receptor-Level Stimulation
Dopamine agonists directly stimulate D2/D3 receptors independent of presynaptic neurons. Examples include:
Pramipexole
Ropinirole
Rotigotine
Strategic Role
Used in early PD to delay levodopa initiation
Adjunct in advanced PD to reduce “off” time
Risks
Impulse control disorders
Hallucinations
Sleep attacks
The pharmacodynamic rationale is elegant; the neuropsychiatric consequences demand vigilance.
MAO-B Inhibitors and COMT Inhibitors
To prolong dopamine availability:
Selegiline
Rasagiline
Safinamide
And peripherally:
Entacapone
Opicapone
These agents extend levodopa’s half-life and smooth motor fluctuations.
They exemplify pharmacokinetic optimization—refining delivery rather than redefining mechanism.
II. Managing Motor Complications: From Pulsatility to Continuity
The pulsatile stimulation hypothesis suggests that fluctuating dopamine levels drive dyskinesias. Thus, modern strategies aim for continuous dopaminergic stimulation.
Controlled-Release Formulations
Extended-release levodopa formulations reduce plasma variability.
Levodopa–Carbidopa Intestinal Gel (LCIG)
Continuous jejunal infusion stabilizes dopamine levels in advanced disease.
Amantadine for Dyskinesia
Amantadine reduces levodopa-induced dyskinesia via NMDA antagonism.
Here, glutamatergic modulation intersects with dopaminergic therapy—illustrating the network complexity of basal ganglia physiology.
III. Surgical Neuromodulation: Deep Brain Stimulation
The Neurological Engineering Revolution
When pharmacology reaches its limits, neuroengineering intervenes.
Deep brain stimulation (DBS) involves stereotactic implantation of electrodes in:
- Subthalamic nucleus (STN)
- Globus pallidus interna (GPi)
High-frequency electrical stimulation modulates pathological firing patterns.
Indications
- Severe motor fluctuations
- Medication-refractory tremor
- Dyskinesia
Benefits
- Reduction in “off” time
- Decreased medication requirement
- Improved motor function
DBS represents a shift from molecular therapy to circuit therapy—an evolution from chemistry to bioelectric modulation.
IV. Non-Motor Symptom Management: The Silent Burden
Parkinson’s disease extends beyond movement. Depression, cognitive decline, constipation, sleep disturbance, and autonomic dysfunction often precede motor symptoms.
Depression
Selective serotonin reuptake inhibitors (SSRIs) and SNRIs are commonly used.
Psychosis
Low-dose Clozapine or Quetiapine may be employed cautiously.
Dementia
Rivastigmine improves cognition in PD dementia.
Treatment must therefore be multidimensional, integrating neurology, psychiatry, and internal medicine.
V. Emerging Disease-Modifying Strategies
1. Alpha-Synuclein Targeting
Immunotherapy aimed at α-synuclein aggregation is under investigation. The objective: halt propagation of pathological protein spread.
2. Gene Therapy
Viral vector–mediated delivery of aromatic L-amino acid decarboxylase (AADC) to enhance dopamine synthesis.
3. Stem Cell Therapy
Transplantation of dopaminergic progenitor cells seeks true neurorestoration.
Ethical, immunological, and regulatory complexities remain substantial—particularly in countries with variable oversight.
4. Neuroprotective Agents
Agents targeting mitochondrial dysfunction, oxidative stress, and inflammation are under evaluation.
However, definitive disease-modifying therapy remains elusive.
VI. Lifestyle, Rehabilitation, and Integrated Care
Pharmacology alone is insufficient.
Exercise
Aerobic and resistance training improve motor function and may promote neuroplasticity.
Physiotherapy
Improves gait and posture.
Speech Therapy
Addresses hypophonia and dysarthria.
Nutrition
Protein redistribution diets may optimize levodopa absorption.
Holistic care is not complementary—it is essential.
VII. Precision Medicine and the Future
Genetic mutations such as LRRK2 and GBA open avenues for targeted therapy. Biomarker discovery—CSF α-synuclein, neurofilament light chain, and imaging markers—may allow early intervention.
Digital therapeutics, wearable sensors, and AI-driven monitoring are redefining longitudinal care.
The next frontier is not symptomatic control but preclinical interception.
Strategic Implications for India
India faces unique challenges:
- Delayed diagnosis
- Limited access to movement disorder specialists
- High out-of-pocket expenditure
- Variable availability of DBS centers
Policy integration is necessary:
- Strengthening neurology training programs
- Expanding tertiary care infrastructure
- Promoting indigenous device manufacturing
- Encouraging neuropharmacology research
As India moves toward pharmaceutical sovereignty, investment in neurodegenerative research must become a national priority.
Conclusion: From Dopamine Replacement to Neural Restoration
The therapeutic evolution of Parkinson’s disease mirrors the evolution of neuroscience itself.
We began with dopamine replacement.
We progressed to receptor modulation.
We engineered circuits.
We now aspire to regenerate neurons.
Yet the ultimate goal remains unchanged:
To preserve human dignity in the face of neurodegeneration.
Parkinson’s disease treatment is not merely about controlling tremor—it is about restoring autonomy, stabilizing identity, and defending the neurological architecture of personhood.
The future will not belong solely to levodopa.
It will belong to integrated, precision-guided, neurorestorative strategies.
And when disease modification finally becomes reality, Parkinson’s disease will transition from an inexorable decline to a manageable neurological condition—perhaps even a preventable one.
That transformation will not be accidental.
It will be scientific.
It will be strategic.
And it must be ethical.
